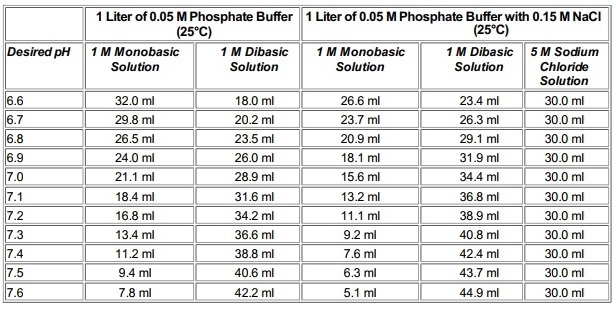
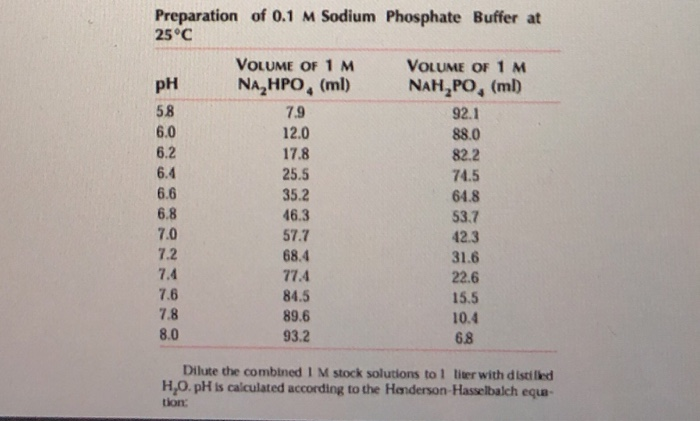
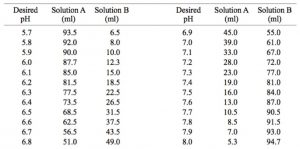
SOLVED: A phosphate buffer solution is prepared by dissolving 25.0 g of potassium hydrogen phosphate (K,HPO ; FM = 174.18) and 12.0 g of potassium dihydrogen phosphate (KH,PO : FM = 136.09)

Preparation of Buffers - 1 Calculate the volume of sulfuric acid (H 2 SO 4 ) necessary to prepare 600 milliliter 0.5M H 2 SO 4 from concentrated H 2 SO. - ppt download
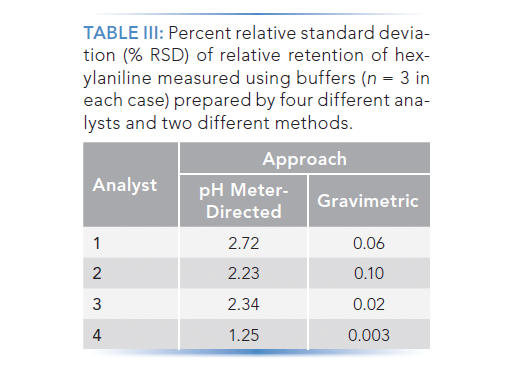
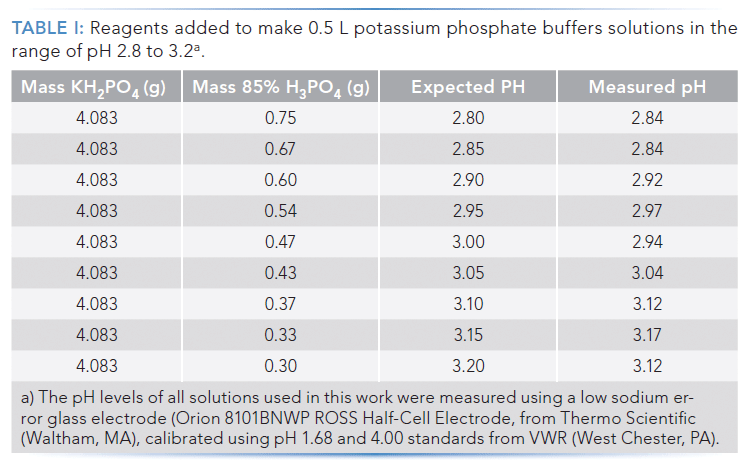
Mobile Phase Buffers in Liquid Chromatography (LC): Effect of Buffer Preparation Method on Retention Repeatability

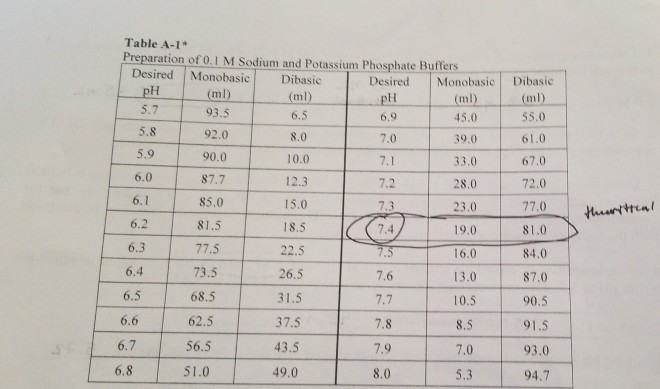
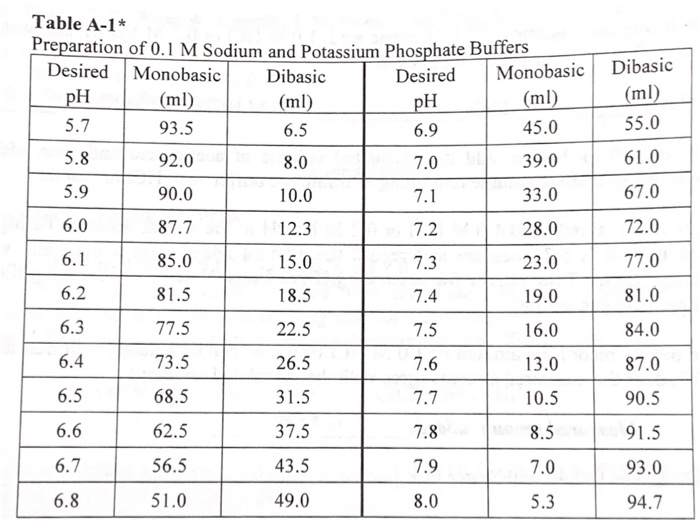
SOLVED: PART B Preparatlon of a two-component dihydrogen phosphate hydrogen phosphate buffer You will prepare 250mL of O.1M potassium phosphate buffer, pH from solid K-HPO (potassium hydrogen phosphate solid KHPO (potassium dihydrogen