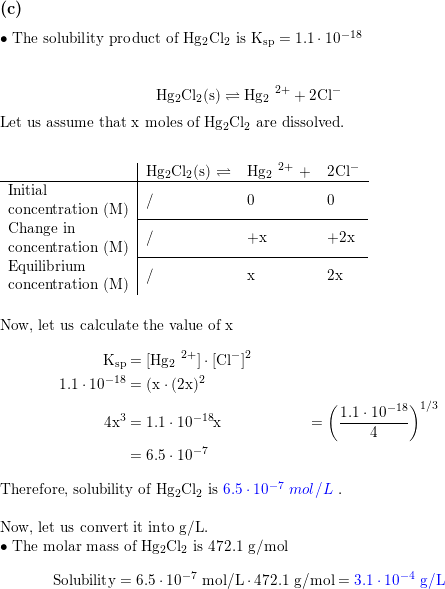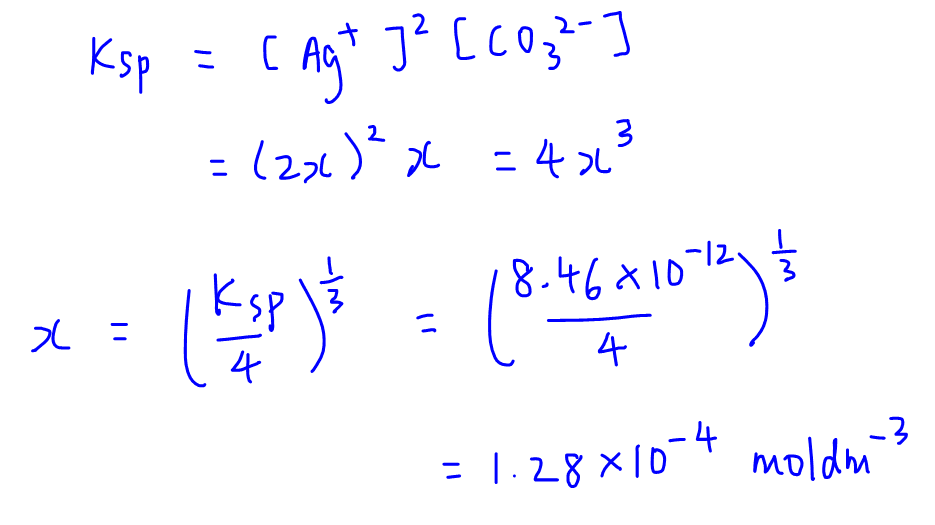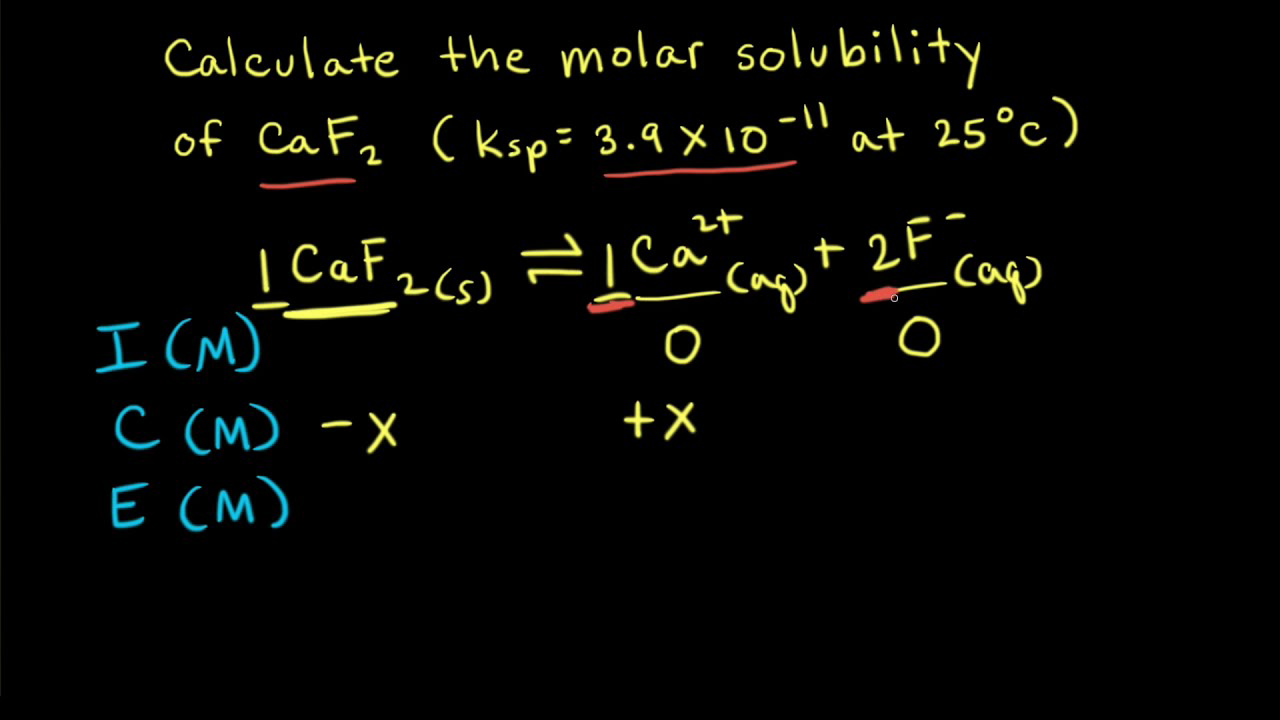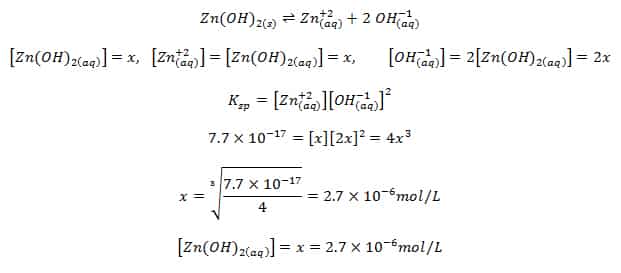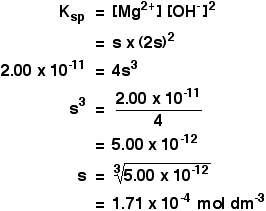Calculate solubility (in moles / litre) of a saturated aqueous solution of Ag3PO4 if the vapour pressure of the solution becomes 750 torr at 373 K(Assume molality = molarity).
![Calculate solubility of AgCN (K(sp)= 4 xx 10^(-16)) in a buffer solution of PH=3. [(Ka)(HCN)= 4 xx 10^(-10)]. Calculate solubility of AgCN (K(sp)= 4 xx 10^(-16)) in a buffer solution of PH=3. [(Ka)(HCN)= 4 xx 10^(-10)].](https://d10lpgp6xz60nq.cloudfront.net/web-thumb/11037183_web.png)
Calculate solubility of AgCN (K(sp)= 4 xx 10^(-16)) in a buffer solution of PH=3. [(Ka)(HCN)= 4 xx 10^(-10)].